StudyIQ is India’s Best Website for UPSC, State PSC, and Judiciary exam preparation. We offer a wide array of resources designed to help aspirants succeed in these highly competitive exams. Our platform provides UPSC Online Coaching, Daily Current Affairs, Government Schemes Updates, General Knowledge Updates, and State PSC Updates, ensuring that you stay well-informed and prepared at all times.
We cover a diverse range of subjects, including History Updates, Geography Updates, Indian Polity Updates, Environment Updates, Science Tech Updates, Economy Updates, and Art and Culture Updates. Our Biography Updates help you learn about influential figures that are often tested in various exams.
Trending Posts
- SSC MTS Apply Online for 1075 Posts – Apply Now at ssc.gov.inJuly 5, 2025Staff Selection Commission (SSC) has officially announced the SSC MTS Online Form 2025 for 1075 Havaldar posts ...
- Dynamic Pricing: What It Is and Why It’s ImportantJuly 5, 2025Context: According to the new Motor Vehicle Aggregator Guidelines (2025), aggregators shall be permitted to charge a ...
- BHARAT Initiative: Establishing Healthy Ageing ParametersJuly 5, 2025Context: The BHARAT study, launched by IISc Bengaluru, aims to identify India-specific biomarkers for healthy ageing and ...
- New Garcinia Kusumae Species Found in AssamJuly 5, 2025Context: Botanists have identified a new species of Garcinia in Assam and named it Garcinia Kusumae in ...
- SSC MTS Previous Year Question Paper with Solution, Download PDFJuly 5, 2025Staff Selection Commission conducts the Multi-Tasking Staff annually to recruit candidates for General Central Service Group-C non-gazetted, ...
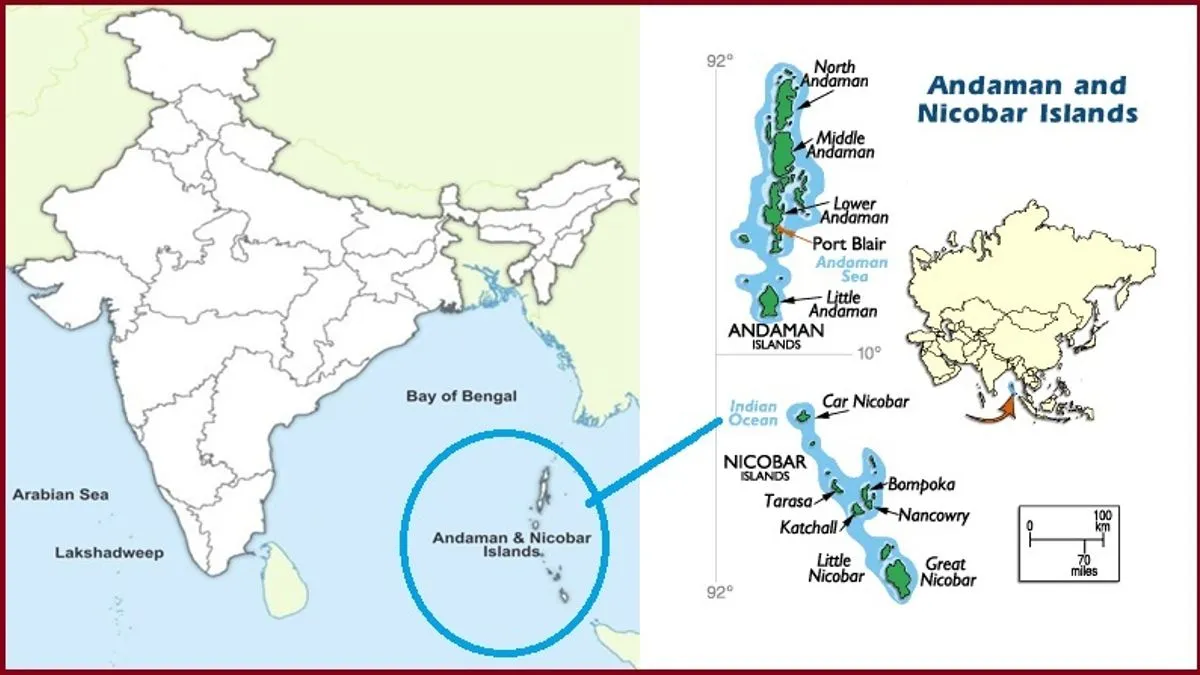
- Andaman Energy Exploration, Aim and SignificanceJuly 5, 2025Context: India is actively exploring vast offshore areas near the Andaman Islands, with early indicators showing potential ...
- SSC MTS Syllabus 2025, Check MTS Exam Pattern and Download PDFJuly 5, 2025The Staff Selection Commission (SSC) has published the SSC MTS 2025 Notification for Multi-Tasking (Non-Technical) Staff and ...
- Sheesh Mahal in Delhi Restoration, Architectural Style and Historical SignificanceJuly 5, 2025Context: The historic Sheesh Mahal (Palace of Mirrors) in Delhi has been restored and reopened to the ...
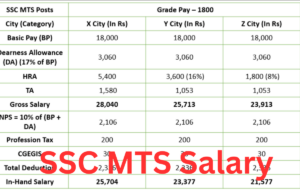
- SSC MTS Salary 2025, Check Highest Salary, Perks and AllowancesJuly 5, 2025The SSC MTS (Staff Selection Commission Multi-Tasking Staff) exam is a significant opportunity for candidates seeking stable ...
Government Schemes
- BHARAT Initiative: Establishing Healthy Ageing Parameters
- C-FLOOD Platform – A Unified Inundation Forecasting System
- ESIC Launches SPREE 2025 to Expand Social Security Coverage
- Employment Linked Scheme (ELS), Objectives and Focus Sector
- BhashaSetu: Bridging Language Barriers with AI Translation in India
- Ek Bharat Shreshtha Bharat Abhiyan – Fostering Unity in Diversity
History
- Maharashtra’s Mendrachi Vancharai System
- Keeladi Excavation: Uncovering the Ancient Tamil Civilization
- Sanyasi Revolt, Objectives, History, Reason and Important Leaders
- Indus Valley Civilization, History, Phases, Script and Significance
- Pandyan Dynasty, Administration and Social and Political Aspects
- Regulating Act of 1773: Background, Provisions, Significance & Drawbacks
Geography
- Places in News for UPSC 2025 for Prelims and Mains Exam
- Iron Ore, Types, Uses, Distribution in India and in World Map
- Low Pressure System and Madden-Julian Oscillation (MJO)
- Desert Climate, Distribution, Climatic Conditions & Map
- Largest Desert in the World, Check Name, Location, Type, Area
- Deserts of India Map, Features of Thar Desert & Cold Desert of India
Indian Polity
- Indian Secularism: Constitutional Provisions and Associated Challenges
- Special Intensive Revision (SIR) of Electoral Rolls
- India Mediation Campaign, Objectives, Process and Significance
- Emergency Provisions in Indian Constitution: Types and Significance
- Directive Principles of State Policy (DPSP) Meaning, Articles, Features, Importance
- National Company Law Appellate Tribunal (NCLAT)
Environment
- New Garcinia Kusumae Species Found in Assam
- Andaman Energy Exploration, Aim and Significance
- Aravallis Degradation and Restoration: Impact and Proposed Solutions
- Species Added to India’s Flora and Fauna Database
- Salkhan Fossil Park Joins UNESCO’s Tentative List of World Heritage Sites
- Forest Conservation Act 1980, Objectives, Features, Amendments
Economy
- Dynamic Pricing: What It Is and Why It’s Important
- Asset Monetisation Strategy for Road Sector and Its Significance
- World of Debt Report 2025, Key Highlights and Reforms
- Index of Industrial Production 2025, Formula, Calculation, Importance
- List of GI Tag Products in India 2025, State Wise GI Tags
- Sagarmala Finance Corporation Limited (SMFCL): Objectives and Functions
Biography
- Savitribai Phule Biography, Early Life, Contribution and Legacy
- Tansen Biography, Musical Legacy and Master of Dhrupad
- Sree Narayana Guru (1856–1928), Key Movements and Literary Works
- Mangal Pandey Biography, History and Role in Revolt of 1857
- Dr. Shyama Prasad Mukherjee Biography, Legacy, and Contributions to Indian Politics
- Meet Saraswathi Rajamani: India’s First Female Spy during Freedom Struggle
Best UPSC IAS Preparation Website 2025
The objective of the StudyIQ is to provide every student across the country with affordable resources that can help them in their preparation for UPSC, State PSC and Judiciary exams.
StudyIQ covers all the recruitment information from the start till the end. After the release of the notification of the exam, we cover the detailed information that a candidate wants to know about a particular recruitment, such as the notification, important exam dates, vacancy details, eligibility criteria, exam pattern, syllabus, and educational qualifications. We also provide you with a direct link to download the notification PDF directly and steps to download or apply for the particular examination from the respective official website.
StudyIQ provided appropriate study material with the test series, book kit, mock test, previous years’ question papers with solutions, cut off, and daily MCQs. We provide you with vast knowledge of all subjects required for examination, section-wise knowledge, and also videos on the daily relevant current affairs topics on our StudyIQ YouTube platform.
Our test series, daily study notes/PDF, and mock tests help candidates prepare themselves on a daily basis for the UPSC, State PSC, and Judiciary examinations. This will help candidates increase their speed and Accuracy without going to any of the big cities for preparation.
Candidates can attempt proper section-wise mock tests for all topics at their home comfort zone. We also provide daily MCQs for a particular topic of the subject.
As we see there are ample amounts of study materials available in the market that sometimes dilute the focus of the candidates from the focused area. So, the StudyIQ team provides you with all relevant information in one place in a systematic way that can help you figure out the strategy and planning to complete their syllabus in a timely manner.
Best Website for UPSC Preparation 2025
The Union Public Service Commission conducts the UPSC CSE exam every year for the recruitment of various vacancies for the various posts of Group A and Group B under the central government. UPSC Civil Service 2025 is the biggest and most prestigious government examination. StudyIQ provides you with detailed information regarding the notification, exam pattern, eligibility criteria, important dates and syllabus, and Results.
Best UPSC Civil Service Preparation Website FAQs
Q. How do I start my UPSC preparation?
Ans. Candidates can start their UPSC preparation from the StudyIQ platform in a systematic way where you can find Daily Current Affairs, Burning Issues, Hindu Analysis, PIB Analysis and many more.
Q. How to cover current affairs when the IAS preparation syllabus is so vast?
Ans. Current Affairs for the IAS preparation is a very important part for which you can refer the StudyIQ’s Daily Current Affairs news Analysis and monthly magazine.
Q. Is self-preparation for the IAS exam enough to clear it?
Ans. Self-preparation for the IAS exam is a good choice but you need to start preparation in an organized manner with relevant study materials and proper strategy for which the StudyIQ platform can play an important role to provide all the UPSC-related materials and guidance at a place.
Q. Where to download UPSC study materials/IAS study material PDF?
Ans. Candidates can easily download UPSC study materials/IAS study material PDFs from India’s best UPSC platform StudyIQ.
Q. What are the Study Materials for GS preparation for IAS?
Ans. The Study Materials for GS preparation for the IAS exam include mainly NCERT from classes 6th to 12th, Daily Current Affairs & News analysis integrate with the static part and also refer to StudyIQ’s reference books such as Indian Geography, Indian Polity, Indian Economy, Indian Art & Culture and Fundamentals of Geography.
Q. Which website is best for the UPSC IAS exam preparation?
Ans. StudyIQ is the best platform for every aspect of the UPSC IAS exam preparation from scratch.