Table of Contents
Key Findings of the Scientific Assessment
- Ozone layer recovery: If current policies continued to be implemented, the ozone layer is expected to recover to 1980 values by 2066 over Antarctica, by 2045 over the Arctic, and by 2040 for the rest of the world.
- What led to the recovery?
- The steady improvement of the ozone layer since the 2000s is mainly attributed to the effective implementation of the Montreal Protocol of 1989.
- The implementation of the protocol led to the successful elimination of some harmful industrial chemicals, together referred to as Ozone Depleting Substances or ODSs
- The assessment has reported that nearly 99 per cent of the ODSs banned by the protocol have now been eliminated from use, resulting in a slow but definite recovery of the ozone layer.
- ODSs and Climate Change: The report mentioned that the elimination of ODSs has an important climate change co-benefit as well.
- Considering that these substances are also powerful greenhouse gases, the report said that global compliance with the Montreal Protocol would ensure the avoidance of 0.5 to 1 degree Celsius of warming by 2050.
What is the Ozone Layer and its Significance?

- The ozone layer or ozone shield is a region of Earth’s stratosphere that absorbs most of the Sun’s ultraviolet radiation.
- Location: It is mainly found in the lower portion of the stratosphere, from approximately 15 to 35 kilometers above Earth’s surface, although its thickness varies seasonally and geographically.
- Composition: The Ozone layer is made up of Ozone (chemically, a molecule having three Oxygen atoms, or O3).
- The ozone layer contains less than 10 parts per million of ozone, while the average ozone concentration in Earth’s atmosphere as a whole is about 0.3 parts per million.
- Ozone formation: The production of ozone in the stratosphere results primarily from the breaking of the chemical bonds within oxygen molecules (O2) by high-energy solar photons.
- This process, called photodissociation, results in the release of single oxygen atoms, which later join with intact oxygen molecules to form ozone.
- Rising atmospheric oxygen concentrations some two billion years ago allowed ozone to build up in Earth’s atmosphere, a process that gradually led to the formation of the stratosphere.
- Significance of Ozone Layer:
- It effectively blocks almost all solar radiation of wavelengths less than 290 nm from reaching Earth’s surface, including certain types of ultraviolet (UV) and other forms of radiation that could injure or kill most living things.
- Ozone absorbs more than 99 per cent of UV-C rays — the most dangerous portion of the spectrum.
- Ozone absorbs about 90 per cent of the UV-B rays — but the 10 per cent that makes it through is a big factor in inducing sunburns and triggering skin cancer.
- Ozone absorbs about 50 per cent of the UV-A rays.
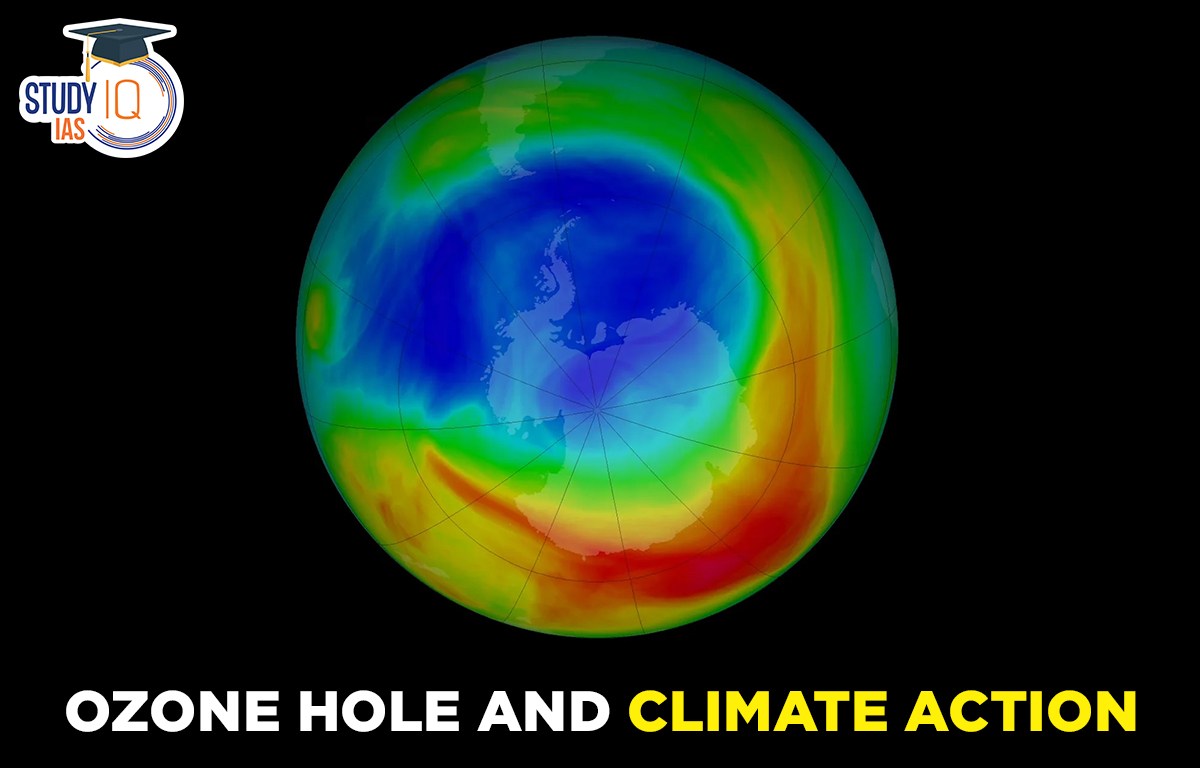
Ozone Layer Depletion

- The depletion of the ozone layer, first noticed in the early 1980s, used to be the biggest environmental threat before climate change came along.
- Causes of depletion: By the middle of the 1980s, scientists had figured out that the chief cause of ozone depletion was the use of a class of industrial chemicals that contained chlorine, bromine or fluorine.
- Most common of these were the chlorofluorocarbons or CFCs, which were used extensively in the air conditioning, refrigeration, paints, and furniture industries.
- Why the depletion is more pronounced over Antarctica?
- The rapid depletion of ozone over Antarctica is mainly linked to the unique meteorological conditions — temperature, pressure, wind speed and direction — that prevail over Antarctica.
- The ozone hole over Antarctica is the biggest during the months of September, October, and November.
Global Efforts to Prevent the Depletion of Ozone Layer
| Vienna Convention |
|
| Montreal Protocol (under Vienna Convention) |
|

 Daily Quiz 24 April 2026
Daily Quiz 24 April 2026
 Wind Energy in India: Definition, Advant...
Wind Energy in India: Definition, Advant...
 Biotechnology: Potential, Regulatory Cha...
Biotechnology: Potential, Regulatory Cha...






















