Table of Contents
Context: India’s ambitious target to significantly lower TB morbidity and mortality and aim for its elimination by 2025 has stalled. Truenat is a rapid molecular test developed in India for diagnosing pulmonary, extrapulmonary, and rifampicin-resistant tuberculosis.
More In News
- The Indian Council of Medical Research (ICMR) is considering adjustments to the TB treatment protocol, including changes in medication and treatment duration, to revitalise the campaign to eradicate TB.
- Despite over 50 years of TB control efforts, tuberculosis remains the most critical health crisis in India.
- TB is responsible for the deaths of approximately 480,000 Indians each year, which translates to over 1,400 deaths daily.
- Annually, more than a million TB cases in India are ‘missing’ or unreported, with many cases either going undiagnosed or inadequately treated, particularly in the private sector.
- It was recognized at the 77th World Health Assembly in Geneva for its significant role in combating TB and its potential in global healthcare solutions.
- Truenat is the first molecular test recommended by the WHO for TB and rifampicin resistance detection at sites with minimal infrastructure.
About Truenat Molecular Testing
- Developed: By Goa-based Molbio.
- Launched: In 2017
- Features:
- It is a portable, battery-operated real-time quantitative micro-PCR system.
- It can be deployed at labs, health centres, and in the field, delivering results in less than an hour.
- It can test for over 40 diseases using disease-specific chips, including a test for COVID-19.
- Significance: Early diagnosis and treatment are critical in combating TB, which kills an estimated 480,000 Indians annually, with India accounting for 27% of the global TB burden.
Impact
- Truenat has approximately 10,000 installations worldwide, with countries reporting significant improvements in TB case detection.
- In India, Truenat is used at over 7,000 primary and community health centres under the National TB Elimination Programme and at about 1,500 private labs.
- Following approval by its national regulatory authority, India is also using Truenat for decentralised COVID-19 testing, with over 9 million tests supplied so far.
Related Information
- To help countries in planning for adoption and scale-up of the tests, the Stop TB Partnership has also launched a Truenat Implementation Guide, developed jointly with the United States Agency for International Development (USAID) and the Global Laboratory Initiative (GLI).
- Rifampin, also known as rifampicin, belongs to the antimicrobial class of drugs.
- This medication is used to manage and treat diverse mycobacterial infections and gram-positive bacterial infections.
About Tuberculosis (TB)
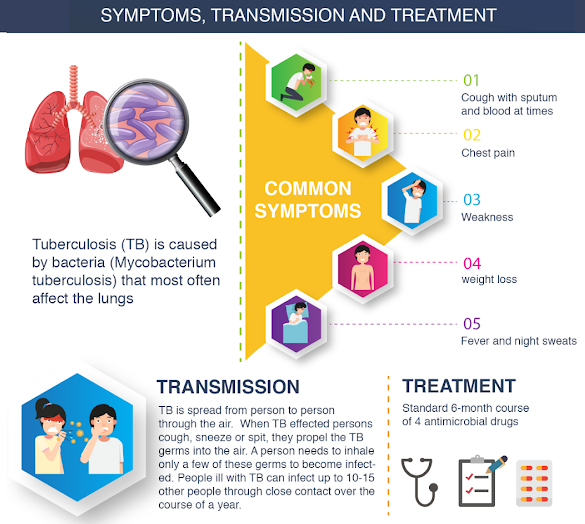
- Tuberculosis (TB) is a bacterial infection spread through inhaling tiny droplets from the coughs or sneezes of an infected person.
- TB is caused by a bacterium called Mycobacterium tuberculosis, belonging to the Mycobacteriaceae family.
- Worldwide, TB is the 13th leading cause of death and the second leading infectious killer after COVID-19 (above HIV/AIDS).
- Transmission: TB spreads from person to person through the air. When people with lung TB cough, sneeze or spit, they propel the TB germs into the air.
- In humans, TB most commonly affects the lungs (pulmonary TB), but it can also affect other organs (extra-pulmonary TB).
- Cure: TB is a treatable and curable disease.
- Vaccine: Currently, Bacille Calmette-Guérin (BCG) is the only licensed vaccine available for the prevention of TB.
-
- BCG works well in some geographic locations and not so well in others.
- Generally, the farther a country is from the equator, the higher is the efficacy.
- It has a high efficacy in the UK, Norway, Sweden and Denmark; and little or no efficacy in countries on or near the equator like India, Kenya and Malawi, where the burden of TB is higher.
- Drug-Resistant TB: Sometimes drug-resistant TB occurs when bacteria become resistant to the drugs used to treat TB. This means that the drug can no longer kill the TB bacteria.
| About Drug-resistant tuberculosis (DR-TB) |
Multidrug-Resistant Tuberculosis (MDR-TB)
Extensively Drug-Resistant Tuberculosis (XDR-TB)
|
WHO Global TB Report 2023
Global Findings
- Tuberculosis was the second leading cause of death from a single infectious agent worldwide in 2022, following Covid-19.
- TB deaths were almost double those from HIV/AIDS.
- Over 10 million people were infected with TB globally in 2022.
- 87% of new TB cases in 2022 were concentrated in 30 countries, with India, Indonesia, China, the Philippines, Pakistan, Nigeria, Bangladesh, and the Democratic Republic of Congo being among the top eight.
- In 2022, 5 million people were diagnosed with TB, the highest since WHO monitoring began in 1995.
- The mortality rate from untreated TB is approximately 50%.
- With current WHO-recommended treatments, about 85% of people with TB can be cured.
- There was a notable recovery in TB diagnosis and treatment in 2022 after two years of disruptions caused by Covid-19.
- India, Indonesia, and the Philippines contributed to over 60% of the global reduction in TB cases.
- The TB incidence rate increased by 3.9% from 2020 to 2022, reversing a long-term declining trend.
Findings Related to India
- Case Fatality Ratio: India’s TB case fatality ratio was 12% in 2022.
- There were 342,000 TB-related deaths in India in 2022, with 331,000 among HIV-negative and 11,000 among HIV-positive individuals.
- India reported 110,000 cases of multidrug-resistant TB in 2022, underscoring the ongoing public health challenge.
Global Initiatives for TB
- Global Fund: It is a worldwide movement to defeat HIV, TB and malaria.
- Global Fund has become the single largest channel of additional money for global TB control.
- Stop TB Partnership: It is a United Nations hosted organisation that takes initiatives to serve the needs of the people, communities, and countries affected by TB.
- It has 1500 partner organisations which include international, non-governmental and governmental organisations and patient groups.
- Global Plan to End TB 2023-2030: It maps out how to end TB as a public health challenge by 2030.
- It focuses on the need to invest in a new TB vaccine and approve it by 2025.
- It shall mobilise a global investment of US$250 billion for the diagnosis and treatment of 50 million people with TB.
- SDG Goal: Ending the TB epidemic by 2030 is among the health targets of the United Nations Sustainable Development Goals (SDGs).
India’s Initiatives for TB
| Scheme | Details |
| Pradhan Mantri TB Mukt Bharat Abhiyaan |
Objectives:
Components:
|
| Ni-kshya Poshak Yojana |
|
| National TB Elimination Programme (NTEP) |
|
| National Strategic Plans for TB |
|
| TB Harega Desh Jeetega Campaign |
|


 BCCI Not Under RTI Act: CIC Ruling, Supr...
BCCI Not Under RTI Act: CIC Ruling, Supr...
 Oral Remarks and Institutional Limits of...
Oral Remarks and Institutional Limits of...
 Hydroelectric Projects in Upper Ganga Ba...
Hydroelectric Projects in Upper Ganga Ba...






















