Table of Contents
Context: Scientists from the CSIR-Institute of Genomics and Integrative Biology, New Delhi, have developed an enhanced genome-editing system that can modify DNA more precisely and more efficiently than existing CRISPR-based technologies.
More in News
- Researchers modified amino acids in FnCas9 that recognize the PAM sequence on the host genome, increasing the binding affinity of the protein to DNA.
- This modification led to a more effective gene editing tool that is also capable of editing regions of the genome that were previously hard to access.
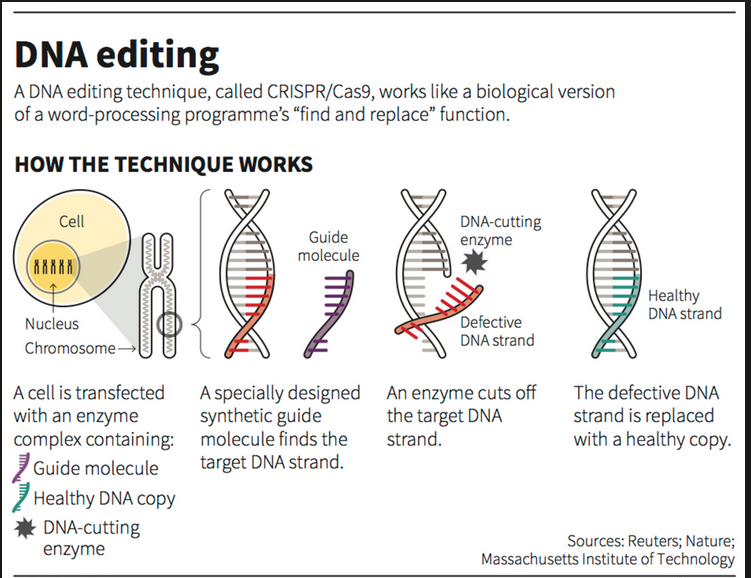
About CRISPR-Gene Editing
- Basis for this technology: Clustered Regularly Interspaced Short Palindromic Repeats (CRISPR), a feature of the bacterial immune system.
- It acts as a genetic memory bank, storing fragments of viral DNA from past infections to bolster future defences.
- When bacteria encounter previously encountered viruses, the CRISPR system enables them to recognize and eliminate these threats, effectively immunising the bacteria.
- Two key components:
- Cas9, an enzyme acting as ‘molecular scissors‘ to cut DNA at specific locations.
- Guide RNA (gRNA), is designed to direct Cas9 to a precise genome spot.
- The gRNA binds to a matching DNA sequence, guiding Cas9 to make strategic cuts.
- Cells then attempt to repair this DNA break, allowing scientists to manipulate genes by adding, removing, or altering DNA segments.
- Applications: Treating genetic diseases, enhancing crop resilience to drought, improving food crop traits, and exploring the potential for bringing extinct species like the woolly mammoth and the dodo back to life.
| Facts |
| CRISPR exists naturally in some bacteria as part of their immune system, identifying and destroying viral DNA. |
CRISPR-Cas9 Gene Editing
- Function: CRISPR-Cas9 allows researchers to add, remove, or alter specific DNA sequences in animal genomes.
- Applications: Used in agriculture to improve plant nutritional value and yield, and in health care for diagnosing and treating genetic disorders.
- Mechanism:
- Guide-RNA (gRNA): Finds and binds to specific genome parts.
- Cas9 Enzyme: Directed by gRNA to the target site, recognizes the protospacer adjacent motif (PAM), and snips damaged DNA.
- DNA Repair: The cell’s repair system inserts the correct DNA sequence.
- Off-Target Effects: CRISPR-Cas9 can sometimes cut unintended genome parts, particularly with the SpCas9 enzyme derived from Streptococcus pyogenes. Engineered versions have higher fidelity but lower efficiency.
- Alternative Enzyme: Researchers are exploring FnCas9 from Francisella novicida bacteria, which is more precise but less efficient.
FnCas9 Advantages
- Enhanced Activity: The modified FnCas9 shows superior activity and precision in cutting target DNA and recognizing single-nucleotide changes compared to its original form.
- Cell Test Results: In laboratory tests involving human kidney and eye cells, the enhanced FnCas9 exhibited better performance and significantly fewer off-target effects.
- Genetic Correction: Enhanced FnCas9 was effectively used to correct a gene mutation responsible for Leber congenital amaurosis type 2 (LCA2), restoring normal RPE65 protein levels in retinal cells derived from patient-specific iPSCs.
- Application in Therapies: The accuracy of the enhanced FnCas9 makes it a promising tool for therapeutic applications, particularly in treating genetic disorders.
Challenges Associated with CRISPR Technology
- Health Equity and Disparities: The potential for CRISPR to exacerbate existing health inequities due to uneven access to advanced treatments across different socioeconomic groups.
- Moral and Ethical Concerns of Germline Editing: The ethical dilemma of making heritable changes to individuals, questioning the morality of altering the genetic makeup of future generations to treat genetic conditions.
- Regulatory Restrictions: Current restrictions limit genome editing to somatic cells, with a moratorium on germline editing due to the complex ethical, societal, and safety concerns.
- Scientific Overreach: Instances like the controversial experiment by Chinese scientist He Jiankui, who edited healthy embryos, highlight the risks of scientists bypassing ethical guidelines and the lack of clear outcomes from such interventions.
- Long-term Effects Uncertainty: The long-term consequences of CRISPR editing are not fully understood, raising concerns about unforeseen impacts on individuals and subsequent generations.
- Accessibility and Affordability: The risk of gene editing technologies becoming exclusive to the wealthy, potentially leading to a societal divide based on genetic enhancements and therapeutic uses.
- Eg. Casgevy costs $ 2.2 million per patient to treat sickle-cell anaemia.
We’re now on WhatsApp. Click to Join
|
Important Terminology |
Germline Editing
Somatic Cells
Sickle-cell Anaemia
|
CRISPR Technology Impact in the Indian Context
Challenges
- Limited healthcare access: Tribal populations suffering from sickle cell anaemia often reside in remote areas with scarce healthcare professionals, hindering primary care and diagnosis.
- Equity and affordability: Concerns exist about wealthy individuals exploiting the technology while marginalised communities lack access due to cost or infrastructure limitations.
Potential Applications
- Sickle Cell Anemia Elimination Mission: Launched in 2023, this government initiative aims to strengthen healthcare systems and improve access to primary, secondary, and tertiary care for affected populations.
- Gene editing as a therapeutic tool: CRISPR could potentially offer curative treatments for sickle cell anaemia, addressing the limitations of current management strategies.
| PYQ |
| Q. What is cas9 protein that is often mentioned in the news? (2019)
(a) A molecular scissors used in targeted gene editing. (b) A biosensor used in the accurate detection of pathogens in patients. (c) A gene that makes plants pest-resistant (d) A herbicidal substance synthesized in generally modified crops Answer: Option (a) |


 Country Strategic Opportunities Programm...
Country Strategic Opportunities Programm...
 LEADS 2025 Report: Key Highlights, Objec...
LEADS 2025 Report: Key Highlights, Objec...
 AI-Powered Financial Inclusion in India:...
AI-Powered Financial Inclusion in India:...






















